 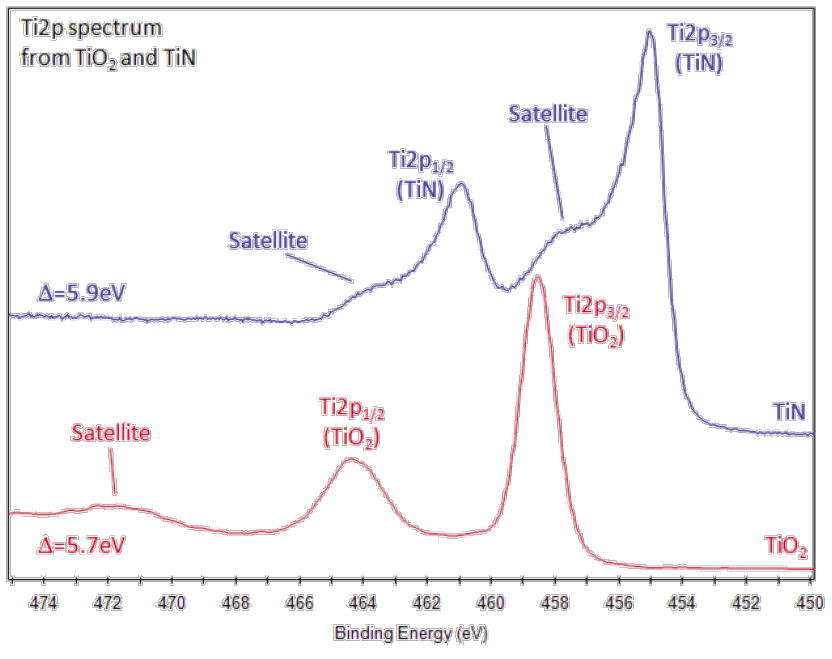
Depth profiles using ion beam etching for elemental compositions as a function of depth in metals and inorganic materials to depths as great as 3 μm, allowing this surface analysis technique to perform sub-surface analysis.Anderson in the early 1990s these complex chemical phases are common on surfaces or interfaces, as they are in minerals Identification and separation of complex chemical phases by differences in surface potentials using a technique developed by Dr.Extensive chemical phase identification based on binding energy shifts characteristic of different bonding configurations and quantitative component analysis.Similarly, the weight ppm is also lower than the atomic ppm for these elements in most glasses and ceramics or even most metal alloys. In weight percent, the detection limit for lead and mercury in most polymers is less than 2 ppm, while that for cadmium is about 3 ppm.Detection limits of 0.001 to 0.005 atomic percent (or 10 – 50 ppm) for heavy metals, 0.03 at.% for lighter transition metals, 0.2-0.4 at.% for O, N, and C, and about 0.4-1.0 at.% for Li, Be, and B.Quantitative elemental analysis of all elements, except hydrogen and helium, which is generally free of matrix effects which degrade quantitative accuracy, unlike Auger Electron Spectroscopy, EDS, XRF, and FTIR.High surface sensitivity, commonly with about 50% of the signal from the outer 6 nm of material, about 70% of the signal from the outer 10 nm, and about 90% of the signal from the outer 20 nm.Determination of their number and energies in spectral peaks allows the measurement of the number and kind of atoms at the surface. The sample is irradiated with low energy, monochromatic aluminum anode x-rays, causing the emission of photoelectrons and Auger electrons.Schematic Diagram of the Monochromatic Small-Spot XPS System at AME 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. Archives
January 2023
Categories |
 RSS Feed
RSS Feed
